Pa. activists hopeful for potential new Alzheimer’s drug

[ad_1]
An estimated 280,000 Pennsylvanians currently live with Alzheimer’s disease.
Phil Gutis is one of them. He was diagnosed with early-onset Alzheimer’s at age 54.
“There were a lot of tears. A lot of tears,” Gutis said on a recent Friday afternoon as he sat on the front porch of his home in New Hope. “But at the same time, there was also a sense of relief, because I knew something was wrong.”
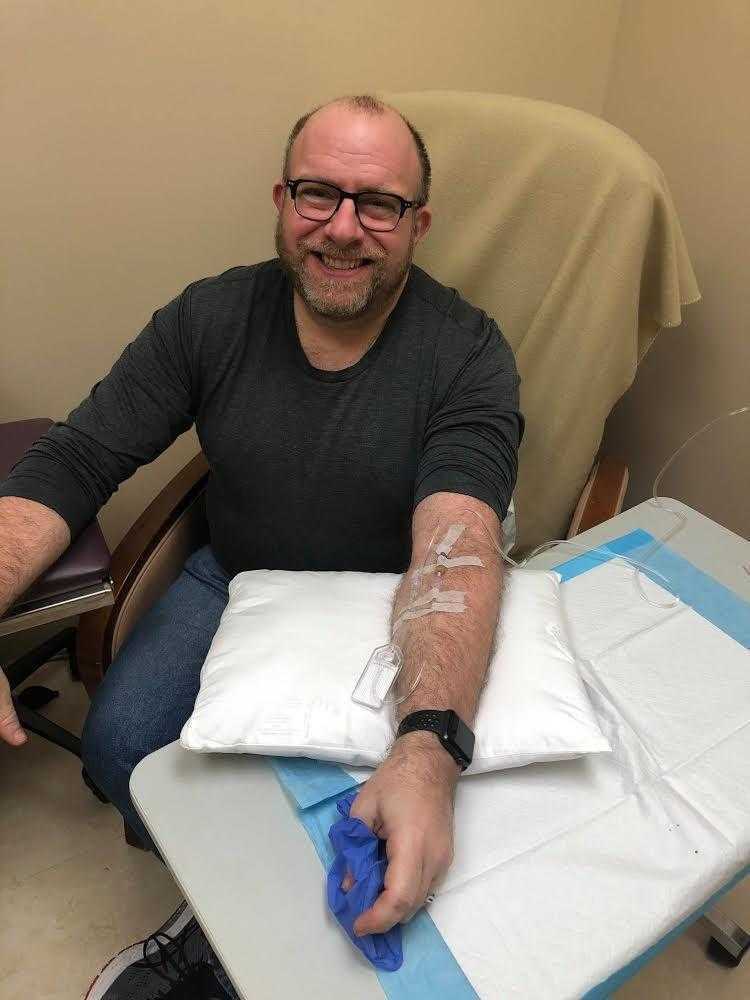
Now six years after his diagnosis, Gutis said he feels good. He continues to work as a freelance editor, writer, activist, and avid LEGO builder. He spends much of his time with his husband, Tim, and their four dogs.
Gutis credits his level of function to medications like Aduhelm, which was controversially approved by the U.S. Food and Drug Administration last year to target and clear away amyloid brain plaque, a biomarker for Alzheimer’s disease.
“The big controversy has been, does that help slow the progression of the disease,” said Gutis, who gets a monthly infusion of Aduhelm. “I think it has.”
Now, a second anti-amyloid drug called lecanemab is showing similar, and possibly clearer, results.
Biotech companies Biogen and Eisai announced last week that preliminary clinical trial data on lecanemab showed early success in slowing cognitive decline up to 27% in people with mild to moderate disease.
More results and data are expected later this year. In an interview Monday with WHYY’s Radio Times, Dr. Jason Karlawish said lecanemab’s results look promising so far.
Notably, Karlawish, a professor of medicine at the University of Pennsylvania and co-director of the Penn Memory Center, said the trial for this drug was more inclusive and better representative of the Alzheimer’s population.
About 25% of participants were Hispanic or African American, according to Biogen and Eisai.
“Compared to other Alzheimer’s trials and frankly, compared to many other diseases of clinical trials, that’s a notable diversity with respect to race and ethnicity,” Karlawish said, “which suggests that efforts to give equal access and improve access to clinical trials for the study were successful.”
[ad_2]
Source link
